Electrolytes as a Test Case
Using battery chemistry to prototype intelligent lab automation
This is not a technical report. It’s not even especially rigorous science. And honestly, that’s part of the point.
At Mirror Physics, we’re building AI tooling to accelerate scientific discovery across both computational and physical workflows. The goal is not to replace the scientist, but to compress the time between an idea, an experiment, and an insight.
Simulation, modeling, and large-scale data analysis are powerful tools we leverage towards this end, but are only a piece of the puzzle. Even with the best computational frameworks, R&D progress hits a wall when research enters the real world. Experiments take time. Iteration is slow. And despite the need for real-world validation, much of this manual work is prone to errors. Luckily for us, recent advances in robotics, additive manufacturing, and agentic control systems make it possible to automate everything from synthesis and sampling tasks to characterization and testing.
It’s surreal to me how much meaningful progress we’ve been able to make as a start-up on real laboratory automation in a short amount of time, with limited resources, and without the kind of infrastructure you’d expect for serious R&D work. The name of the game here is to tackle projects quickly and efficiently.
Why electrolytes?
My own research background is in sustainable materials and clean energy, so when we started asking where AI-driven automation could have the most leverage, climate tech was a natural place to start. I’ve spent enough time doing manual synthesis and characterization to know that if there’s a way to automate monotonous tasks, it’s almost always worth doing.
Our first stop in this broad field of clean energy was battery research, largely driven by conversations I’ve had with colleagues on the importance of innovating energy storage technologies. Specifically, the ion-conducting medium of batteries, electrolytes.
Present-day batteries are fundamentally limited by their electrolytes. Conventional liquid electrolytes were optimized decades ago for conventional graphite-based lithium-ion cells, but they remain flammable, chemically fragile, and poorly matched to emerging battery chemistries. As a result, electrolyte degradation and interphase instability are now among the dominant barriers to higher energy density, faster charging, improved safety, and longer cycle life. Despite their central role, electrolytes are notoriously difficult to design and evaluate. Small changes in composition can dramatically affect transport, stability, and interfacial chemistry, and often in unintuitive ways. This creates a vast, under-explored chemical design space that is poorly suited to manual and low-throughput experimentation.
This is where Mirror Physics comes in.
The high combinatoric design space of electrolytes lends itself well to automation. To demonstrate this potential, we built a simple automated pipeline for electrolyte design, synthesis, and characterization in our Mirror Physics lab. Based in Brooklyn, NY with no infrastructure to handle hazardous chemicals, this lab demo uses sodium sulfate as it’s material of interest. Our initial goal is not to discover the next breakthrough electrolyte chemistry, but to demonstrate the coupling of agentic tooling and automation for a simple but highly generalizable experimental pursuit.
This platform, built in under a month with limited capital, performs automated electrolyte synthesis and characterization. It enables rapid, reproducible exploration of new electrolyte formulations while systematically mapping composition–property–performance relationships and making formulation decisions based on both numerical and empirical data.
Our platform
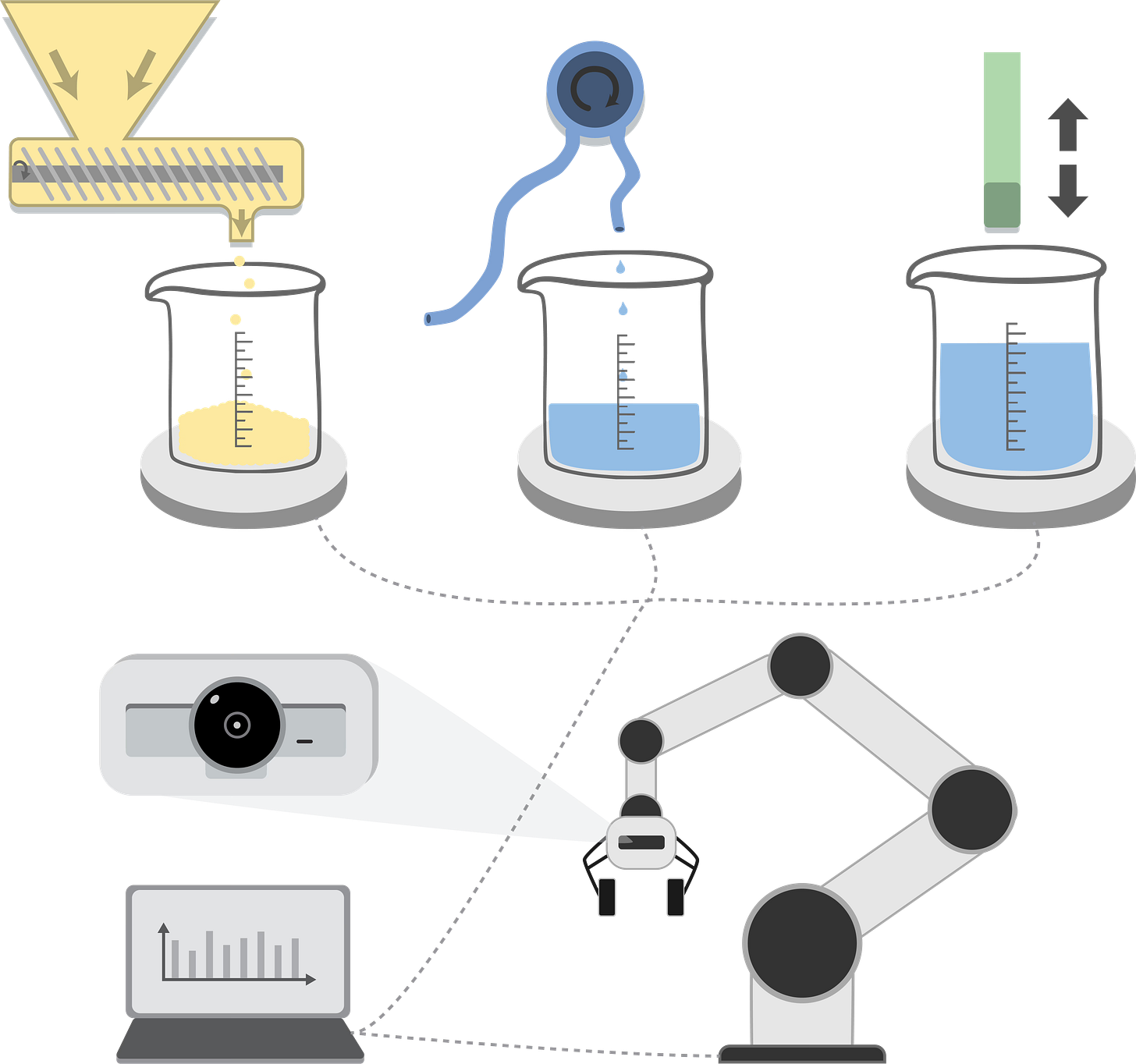
We used 3D printing, off-the-shelf electronics, some miscellaneous lab tools and home-grown code to rapidly prototype three key subcomponents for our electrolytes pipeline: a solid dispensing station, a liquid dispensing station, and a mixing and characterization station. A robotic arm for actuation and object manipulation ties it all together.
Solid dispenser (yellow): A stepper-driven Archimedes screw transports salts from a large hopper to a small outlet, where it falls into a beaker waiting on a digital mass balance. A script processing the balance output regulates stepper actuation using a variable PID control scheme, resulting in a precision of +- 0.01 g per sample.
Liquid dispenser (blue): A peristaltic pump moves liquid from a reservoir to the sample beaker once placed on the 3D printed stage. Liquid deposition is done at a fixed, calibrated rate, where duration is done based on target concentration of the final solution (+- 1 %).
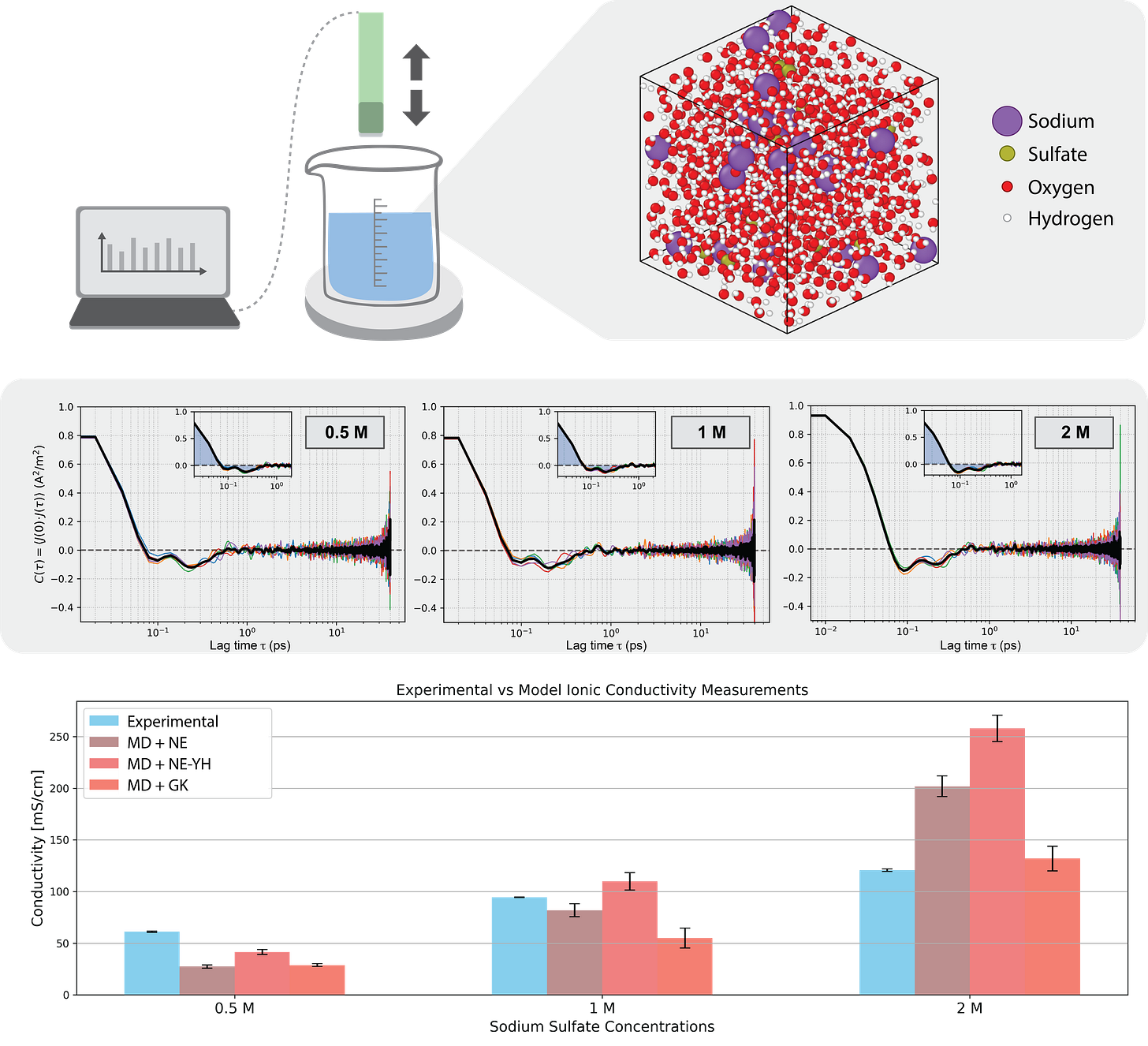
Characterization (green): The final solution is mixed using a magnetic stirring plate, affixed to the skeleton of an Ender 3D printer that makes up the chassis of this stage. An ionic conductivity probe is mounted to the printer toolhead, which is used to probe the solution until measurements reach equilibrium. This information, along with the sample recipe, mixing time, and ambient conditions, are relayed to the central computer for analysis.
Manipulation of the laboratory environment is achieved using a 6-axis robotic arm (UFactory), to which we mounted a web camera. For this demo we use pre-trained motion paths for sample actuation, but progress on the computer vision side could easily enable use of an adaptable robot.
A trial run
The grand question we used to test our automated electrolyte development was “what electrolyte recipe maximizes ionic conductivity?” Now before you say anything, we know this is a trivial use case; we’re limiting the system to sodium sulfate + deionized (DI) water, so it will naturally increase salinity to maximize conductivity. However, it serves as a useful benchmark to illustrate system operation, control, and intelligent feedback.
First, a low salinity solution is made as a starting point. From then on, the system uses recipe information, ambient conditions, and ionic conductivity measurements to decide on the next recipe to test. The “brains” of our lab is a bare-bones Bayesian Optimization (BO) script, connected to our control codes and output to the user via a custom web UI.
We also enabled the lab to run molecular dynamic simulations of each solution to see how close our empirical data was to chemical simulations, which is a bit of a teaser for our new product Axon.
As expected, the lab ran increasingly higher salinity tests until it approached the solubility limit of sodium sulfate in DI water. Each test took about 10 minutes, with some downtime built in as a precautionary measure. Our experiments ran easily, and also matched surprisingly well with conductivity predictions derived from machine-learned interatomic potential (MLIP) molecular dynamic (MD) models via 3 classical methods: Nernst-Einstein (NE), NE + Yeh-Hummer correction (YH), and Green-Kubo (GK). Of these, GK is considered the highest fidelity approach. For those of us less familiar with MD, these MLIP models (run on Axon) are essentially AI-powered simulations of atoms doing their thing, and it’s exciting how close they are to matching with our experiments in the lab.
Final answer? High salinity solution = high conductivity solution. Queue the applause.
Where to next
What excites me most about this demo isn’t the chemistry, it’s the speed enabled by AI-hardware integration. We were able to define a hypothesis, build an experimental system, run closed-loop optimization, and analyze results with minimal human intervention, and without specialized infrastructure or expensive instrumentation. That same pattern applies far beyond electrolytes.
The framework for this intelligent experimentation can be rapidly expanded to cover a broad range of solids, liquids, and characterization methods, while the actuation can increasingly adapt to environmental unknowns via robotics advancements such as force-responsive feedback and computer vision beyond primitive object recognition.
In the lab at Mirror Physics, we’re developing integrated software, hardware, and agentic reasoning capabilities so that experiments can run, think, and act faster, safer, and cheaper than traditional workflows while maintaining transparency and auditability for expert evaluation, intervention, and discovery. Electrolytes are just one concrete example of how this vision is starting to take shape. To be continued!